CTMS
Centralized study planning and execution with real-time visibility, automation, and complete operational control.
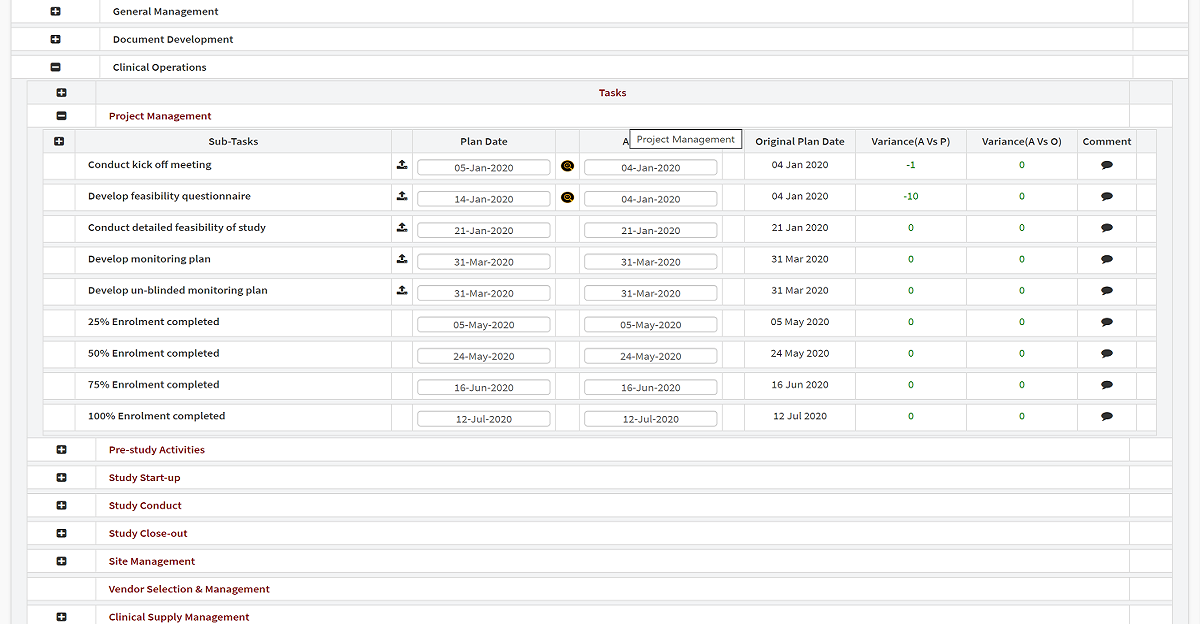
Central Study Planning & Project Management
A completely user-configurable, end-to-end clinical trial management system delivering industry-leading project management capabilities from study inception through close-out.
Advanced analytics and custom workflows provide complete control over every aspect of trial execution — without the need for external consultants.
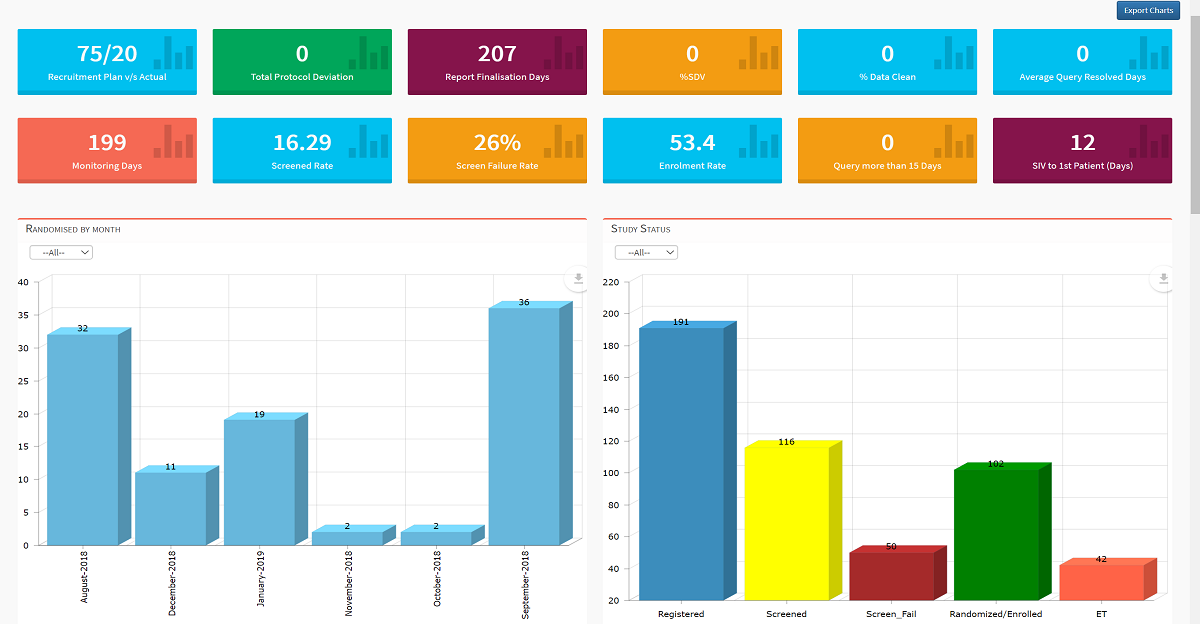
Role-Based Custom Dashboards
User-specific dashboards deliver actionable alerts and insights tailored to Project Managers, Site Monitors, and Sponsor teams.
Collaborative stakeholder canvases provide automated event histories — an industry first — enabling complete transparency, real-time risk visibility, and smarter decision-making.
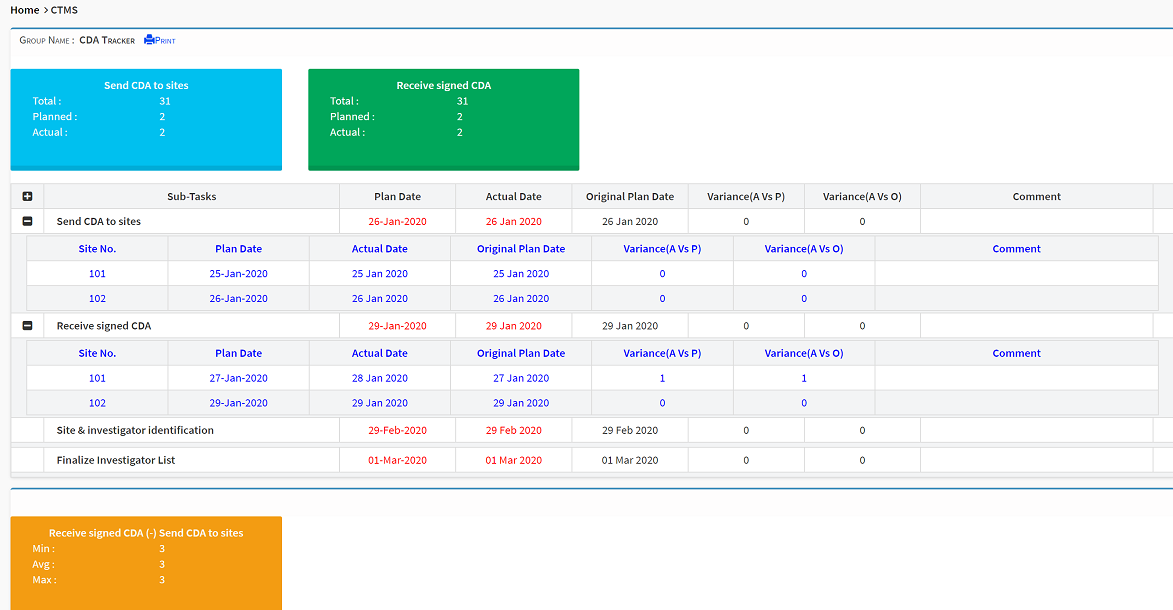
Fully Integrated Workflows
Seamless integration with Site, Deviation, Issue, and Risk Management modules ensures work is done right the first time, improving both time and resource efficiency.
Site visit reports are generated in real time, reviewed, approved, and signed off through a fully automated workflow — and automatically filed in the integrated eTMF.
Real-Time
Site visit reporting & approvals
Role-Based
Dashboards & actionable alerts
Fully Automated
End-to-end CTMS workflows
Run trials with confidence and clarity
Wide Angle Insights CTMS unifies planning, execution, and oversight into a single transparent platform built for modern clinical trials.
Request a Demo