eTMF
Secure, real-time document management delivering inspection-ready trial master files with complete transparency and control.
Trial Documents — Secure, Real-Time Oversight
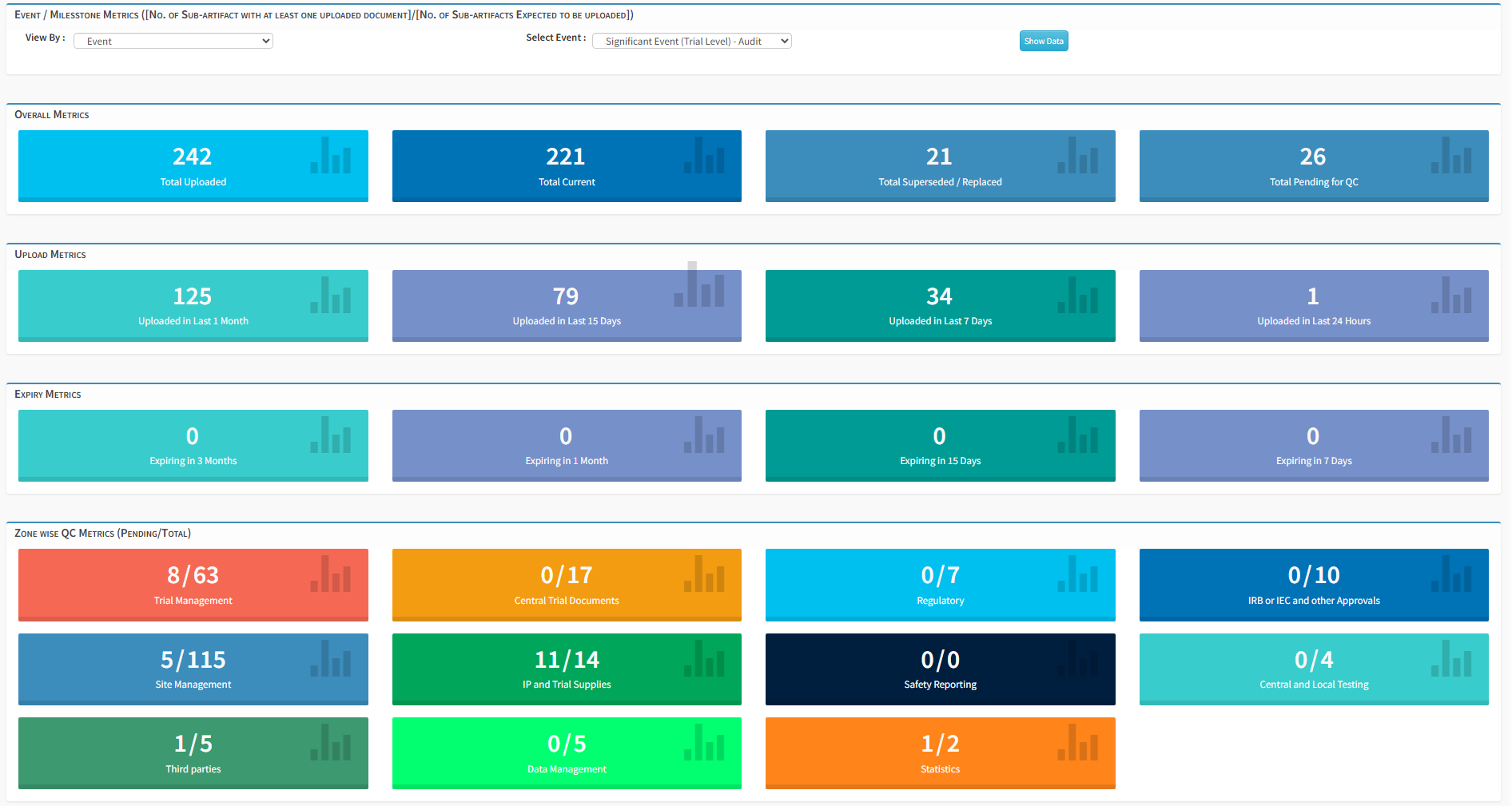
Create, collaborate, review, QC, approve, distribute, and manage all study-related documents throughout the entire lifecycle of a clinical trial.
Documents are available in real time with complete version history, standardized nomenclature, and transparent audit trails — always maintained in an inspection-ready state with role-based, secure access.
Custom Workflows on a Collaborative Canvas
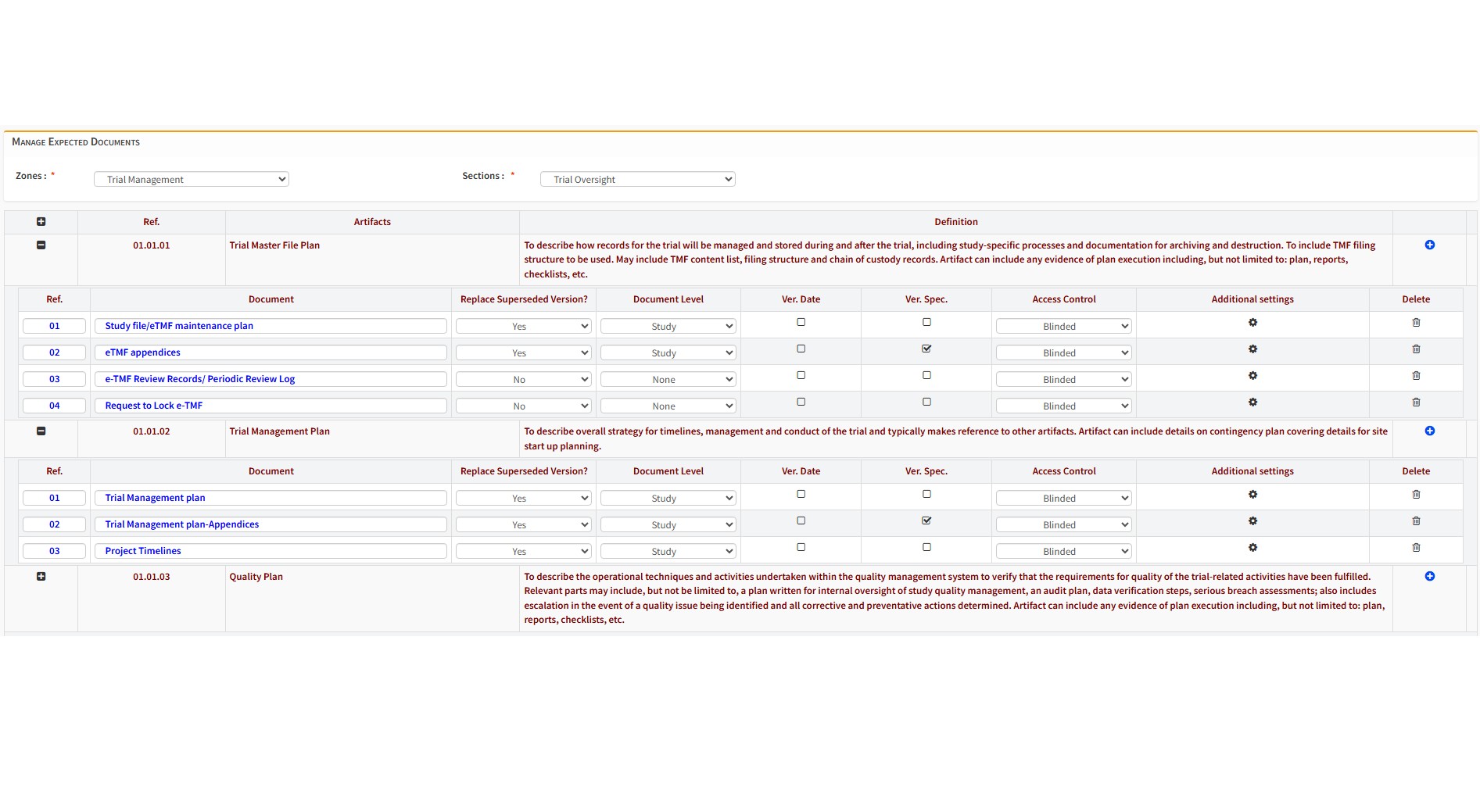
Define workflows tailored to your organization’s processes and track document progress end-to-end using configurable dashboards and real-time metrics.
Sponsors, sites, and CRO teams collaborate transparently, while alerts and notifications highlight delays, QC findings, and workflow bottlenecks early — ensuring timely completion.
Inspection-Ready
At every stage of the trial
Role-Based
Secure document access & audit trails
Collaborative
Sponsor, CRO & site transparency
Document control without complexity
Wide Angle Insights eTMF delivers a modern, collaborative document management platform that keeps your trial inspection-ready at all times.
Request a Demo