Integrated Data Sciences Platform
A next-generation clinical research platform built to unify data, accelerate insight, and proactively manage risk in real time.
Background
During the development of new medicines, vast amounts of research data are generated, reviewed, analyzed, and reported. This data is captured and tracked across multiple, often disparate systems that rarely integrate or communicate with one another.
Data review is performed by diverse teams — medical, data management, biostatistics, and clinical operations — often at different points in time. This lack of real-time coordination contributes to delayed identification of risks to data quality, data integrity, and most importantly, patient safety.
These challenges are amplified by increasing trial complexity, geographic dispersion, variability in investigator experience, site infrastructure limitations, and inconsistent standards of care across regions.
Ensuring the fundamental principles of clinical research are upheld at all times using integrated technology
Patient Safety · Patient Rights · Data Integrity · Data Quality · Transparency of Conduct
Limitations of the Conventional Approach
Traditional approaches to risk management and patient safety are often reactive and prone to review delays, increasing the risk of regulatory non-compliance.
While global regulators increasingly encourage the use of data science and advanced analytics in pharmaceutical development, a significant gap remains in fully integrating all data sources and applying medical, statistical, and operational algorithms to identify risk in real time.
Wide Angle Insights — A Next-Generation Platform Focused on Risk
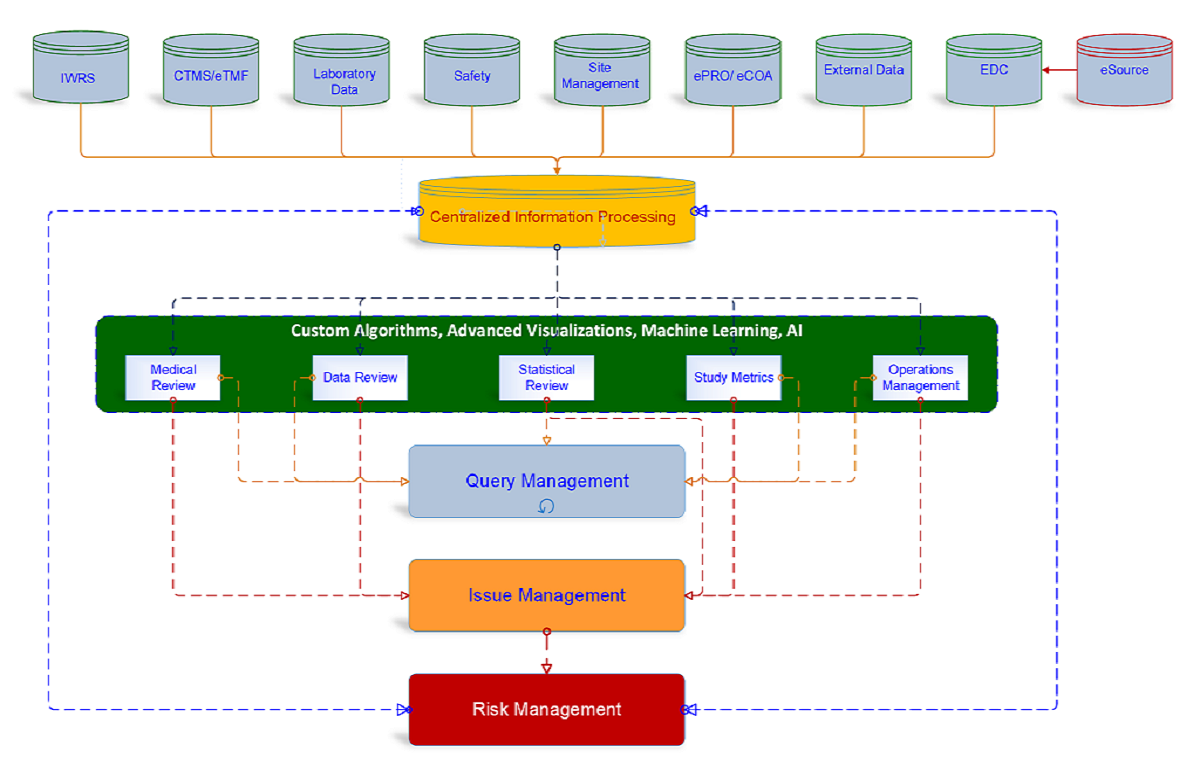
Wide Angle Insights is a fully integrated, source-agnostic data sciences platform that combines intuitive algorithms, smart data reduction, advanced analytics, and visualization to enable real-time signal detection, adjudication, correlation, and risk management.
Built on industry standards such as TransCelerate and ISO 31010, the platform supports collaborative review, transparency, and efficient decision-making across clinical teams.
Conceptualized and developed by experienced clinical research professionals, the platform addresses the shortcomings of siloed, standalone tools and exceeds the capabilities of traditional unified systems.
It enables early risk identification, significant reduction in data errors, and accelerated development timelines — including earlier go/no-go decisions. Predictive analytics and machine learning provide users with full control over clinical trial data, delivering over 75% efficiency gains in resources and timelines.
In addition to standard audit trails, the platform introduces complete, printable, and classified event histories — an industry first. Integrated communication, decision documentation, workflow automation, and high configurability result in measurable efficiency and cost savings.
From fragmented systems to unified intelligence
Wide Angle Insights delivers a truly integrated clinical research platform that transforms data into real-time insight, control, and confidence.
Request a Demo